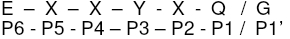
Inhibitors
The cysteine in the catalytic triad is susceptible to cysteine modifying compounds, such as iodoacetamide, leading to a complete inhibition of the TEV protease. Also, 5 mM Zinc is known to inhibit TEV protease. On the other hand 1 mM PMSF, AEBSF or EDTA do not inhibit TEV protease.
Reaction conditions
These are solely guidelines. The optimal cleavage conditions may have to be found empirically.
TEV protease cleaves optimal at pH 8 and at no or low salt.
A typical reaction buffer composition is 50 mM Tris-HCl pH 8.0, 0.5 mM EDTA and 1 mM DTT.
The smeihof TEV protease is usually mixed in a 1:50 to a 1:100 ratio with its substrate.
Alternatively, TEV protease : substrate ratios as low as 1:20 maybe considered although
it is worth noting that even under optimal conditions a TEV protease digestion normally does not exceed 85% efficiency.
The optimal cleavage temperature is 25 -30°C. Results may vary when performing a prolonged digestion at lower temperatures.
On column cleavage is possible but this is usually less efficient.
For on column cleavage it may be helpful to include glycerol or sorbitol in the cleavage buffer.
TEV protease removal after digestion
TEV protease is ~27 kDa in size. Depending on the sizes of your protein and purification tag
it may simply be removed together with the cleaved off purification tag by filtration or dialysis.
Alternatively it may be removed on a nickel NTA column once you removed the DTT and EDTA that may be present in your reaction mixture.
Depending on your application and the cleavage efficiency you may choose to not remove the TEV protease but instead use a low amount of TEV protease relative to the amount of your protein of interest.
Storage
Store at -20°C
Shipment
On ice pack
Troubleshooting
No cleavage
A lack of or insufficient cleavage may have several causes:
1. The protease is inactive.
This typically happens after prolonged storage and/or using sub-optimal storage conditions.
2. The reaction conditions.
High salt and/or extreme pH may inhibit or denature TEV protease.
3. Steric hindrance
The cleavage site may not be accessible to the catalytic site of the TEV protease.
This may be resolved by placing a linker of a few amino acids between the cleavage recognition site and purification tag.
Alternatively the linker may be placed between the cleavage recognition site and protein but this would result in
more foreign amino acids on the protein of interest.
4. Sub-optimal sequence used for the protease recognition site
Check the sequence.
My protein of interest contains disulfide bridges
Try using a redox buffer of glutathione/oxidized glutathione instead of DTT to leave the disulfide bridges intact.
There is a secondary cut site present in your protein of interest. Try shorter incubation times, lowering the digestion temperature or use less protease. Alternatively, there is a contaminating protease present in your reaction mixture.
